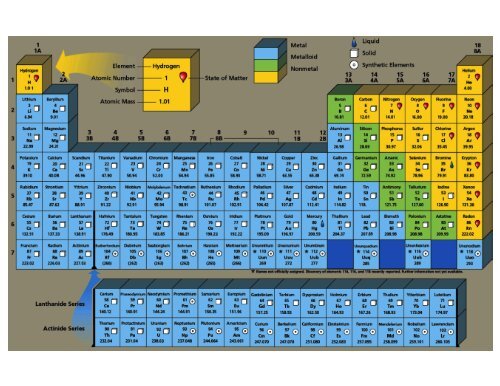
The number of grams of reactants (32.0 g of methane and 128.0 g of oxygen = 160.0 g total) is equal to the number of grams of product (88.0 g of carbon dioxide and 72.0 g of water = 160.0 g total). The answer is (b) Law of conservation of mass.How much hydrogen should be present in a different, 50.0 g same of methane? (Law of Definite Proportions): A sample of methane contains only carbon and hydrogen, with 3.00 grams (g) of carbon for every 1.00 g of hydrogen.(a) What mass of oxygen is needed in this reaction? (b) What mass of oxygen is needed to burn 8.00 lb of wood? 1 lb = 453.59237 g. The products of this reaction weigh 74.4 g. (Law of Conservation of Mass): 36.0 grams (g) of wood are burned in oxygen.Show that these results are consistent with the law of constant composition. 6.92 g of sodium chloride was produced in the second experiment. In the second experiment, 4.20 g of chlorine reacted with 20.00 g of sodium, using up all the chlorine. 11.08 g of sodium chloride was produced in the first experiment. /PeriodicTableCharge-WBG-56a12db23df78cf772682c37.png)
In the first experiment, 4.36 grams (g) sodium are reacted with 32.24 g of chlorine, using up all the sodium. (Law of Definite Proportions) Two experiments using sodium and chlorine are performed. 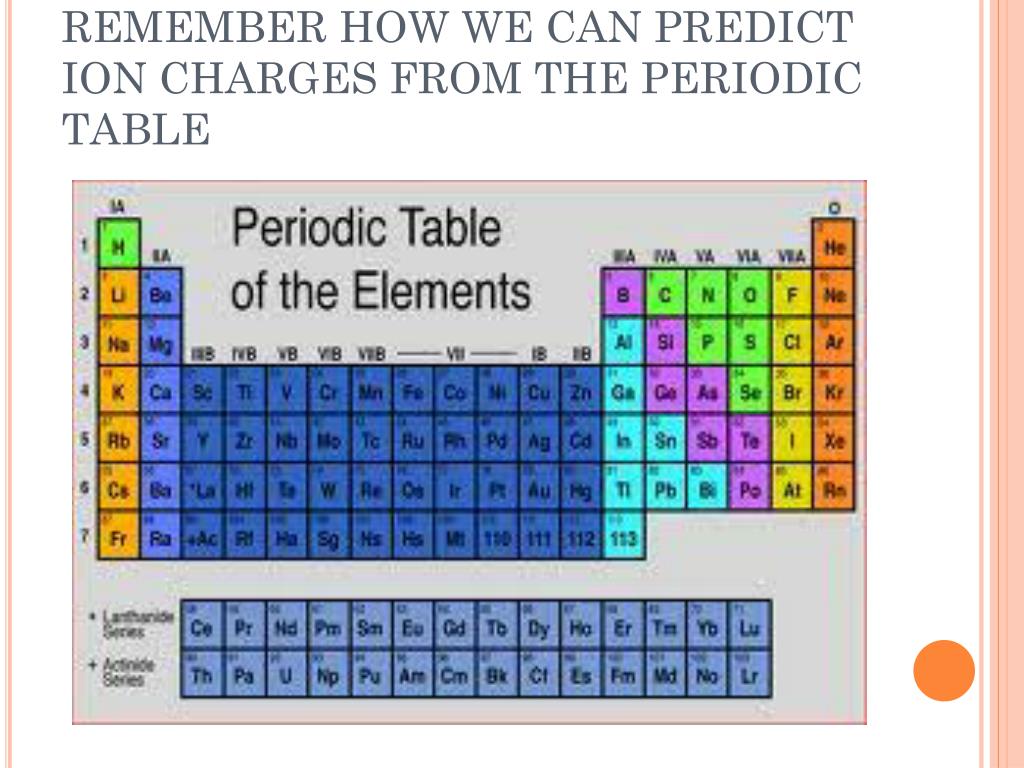
The reaction produces 22.00 g of carbon dioxide and an unmeasured mass of water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
